In recent years, advances in technology and techniques have opened new doors to the recovery of oil and gas from shale formations around the world. The total worldwide available shale oil resources may be equivalent of more than 5 trillion barrels of oil, and the United States is estimated to have more than 3.7 trillion barrels under its lands and controlled waters. Recovering these “unconventional resources” would greatly expand the world’s potential reserves, but recent analysis indicates only 3-10% of this oil is currently economically recoverable.
A small improvement in productivity and recovery could provide access to billions of barrels of recoverable oil.
CO2 injection has been used to improve oil recovery in conventional reservoirs for the last four decades. The injection of this gas into conventional reservoirs has been proven to aid in oil extraction through three primary mechanisms: beneficial oil swelling, reduction of oil viscosity, and reduction of residual oil saturation.
Injecting CO2 in “unconventional” shale oil reservoirs is a relatively new concept and the underlying physical mechanisms are still unknown. Several recent studies suggest CO2 injection could be a viable method to improve recovery for shale oil reservoirs.
However, the majority of these studies are based on assumptions rooted in experience with conventional reservoirs. Datasets and observations from these reservoirs can be helpful, but computer models and our understanding of CO2-oil interactions rely on the behaviors and relationships between the reservoir’s contents and the injection fluids and gases at much larger scales than the pores of shale reservoirs. In reality, these assumptions may not be valid, as we cannot conclude that CO2 and oil interact with each other in the same way in a four nanometer-wide crack as they do in a four micrometer-wide crack.
Dr. Hadi Nasrabadi, an assistant professor and the Douglas Von Gonten Faculty Fellow in the Harold Vance Department of Petroleum Engineering, along with Dr. Debjyoti Banerjee, an associate professor and the Leland T. Jordan Career Development Professor in the Department of Mechanical Engineering, will perform theoretical and experimental studies to evaluate the confinement effects – the impact that smaller spaces have on the properties of substances – that shale reservoirs may present to change our understanding of CO2-oil interactions. Specifically, the pair will analyze how a confined space affects the solubility of CO2 in oil, CO2’s effectiveness in impacting oil swelling, the viscosity reduction of oil as CO2 dissolves, and the overall miscibility of CO2 and oil.
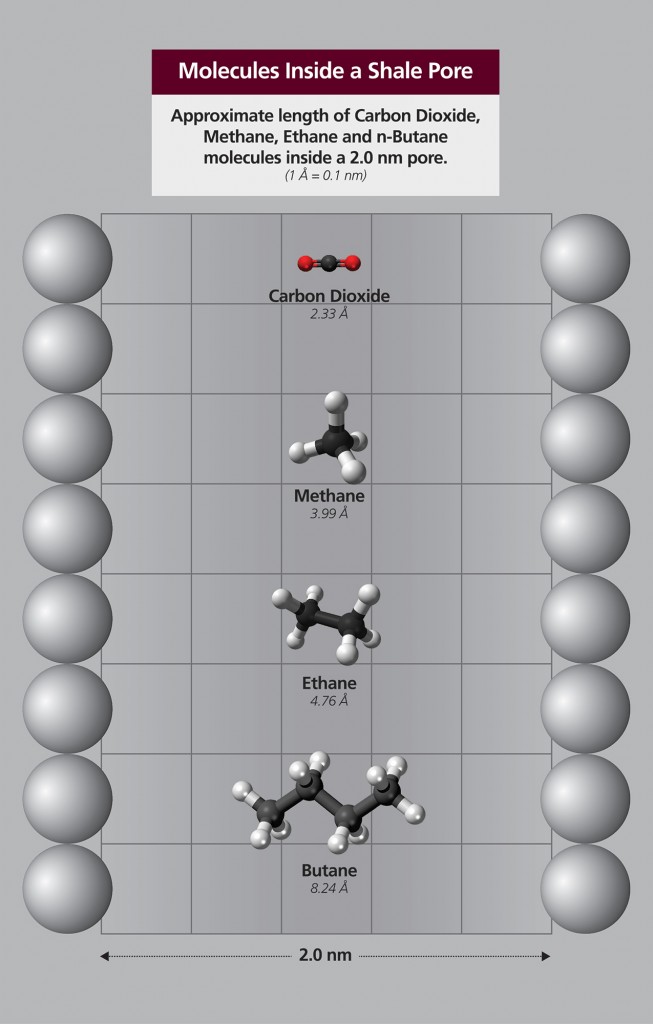
Shale oil reservoirs contain micropores and nanopores with diameters less than two nanometers and some larger pores (mesopores) with diameters from two to 50 nanometers wide. In smaller pores with diameters only a few molecules wide, the surface molecules of the shale may significantly influence the fluid molecules in the pore space. This strong surface-fluid interaction and its competition with fluid-fluid interaction could lead to heterogeneous distribution of fluid molecules throughout the pore space.
Using molecular simulation, Nasrabadi will evaluate the surface-fluid effect on key mechanisms for improved CO2 recovery in shale oil reservoirs. After the simulation, Nasrabadi and Banerjee will compare the simulations with experimental measurements using a microfluidic and nanofluidic device.
Modeling the CO2 as it dissolves into the oil under various conditions – particularly the confined space – will allow Nasrabadi to calculate the nanoscale confinement effects on oil swelling and viscosity reduction and how the varying conditions affect the interactions. Additionally, the modeling will permit the calculation of the change in minimum miscibility pressure, which is the pressure at which the interfacial tension between the injected fluid and oil vanishes, or the point at which the oil and CO2 become a heterogeneous solution.
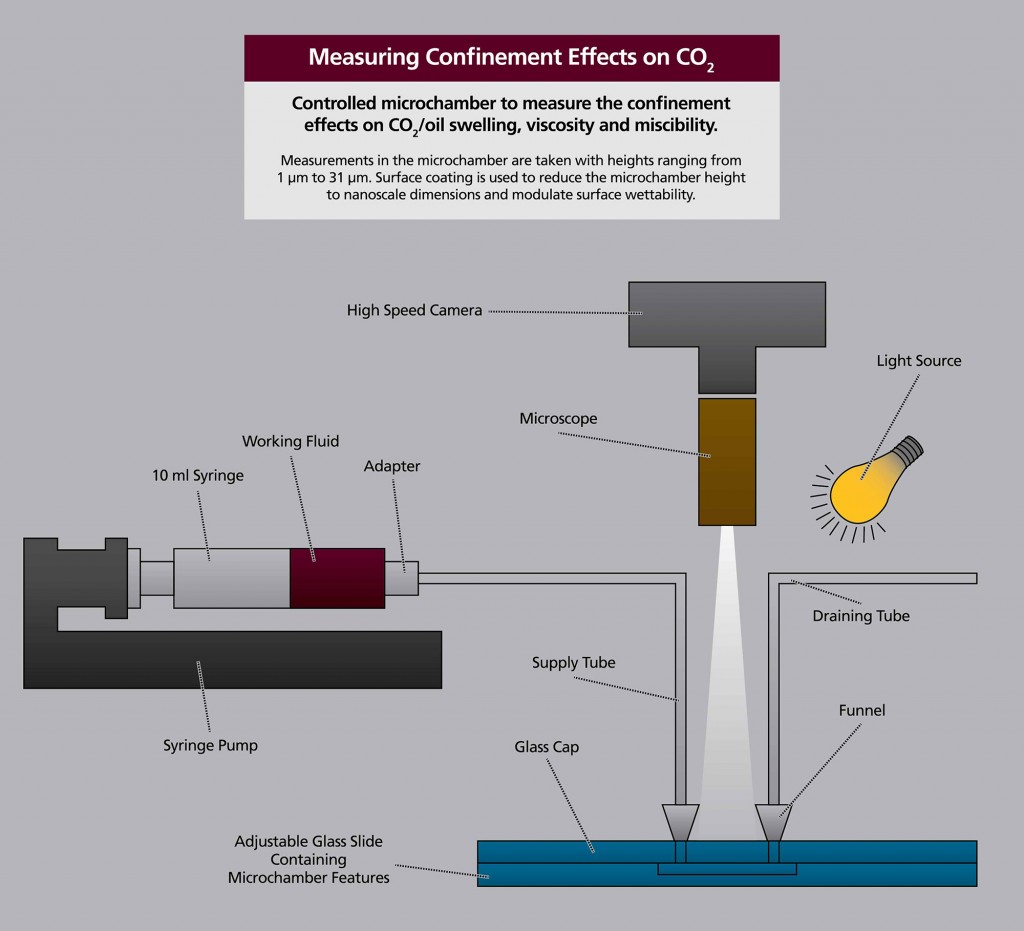
To conduct the experimental measurements, Nasrabadi and Banerjee will construct an adjustable microfluidic and nanofluidic device capable of introducing the CO2 and oil to a glass microchamber with microchannels and nanochannels. Observations will be carried out through a microscope connected to a high-speed camera. Temperature and pressure modifications to the glass microchamber will be controlled, and will allow the pair to find the minimum miscibility pressure.
The overall objective of the project is an increased understanding of the impact of CO2 injections as a method of enhanced oil recovery in shale reservoirs. This knowledge has the potential to provide a framework for developing optimum procedures for recovery, and could lead to significant improvements in recovery of resources currently out of our reach.